 Germanium lies in the p block just under Si, along the diagonal line of semi-metallic elements, which suggests that elemental Ge is likely to have the same structure as Si (the diamond structure). and CH3OH is interpreted as a molecule with one carbon, four hydrogens. Arrange the solids in order of increasing melting points based on your classification, beginning with molecular solids.Ī. Write a rule that will allow you to classify a compound as ionic or covalent on.HF(aq) + NH3(aq) NH + 4 (aq) + F (aq) Net Ionic Equation. The individual melting and boiling points, solubility and other physical properties of molecular compounds depend on molecular polarity. HF(aq) + NH3(aq) NH4F(aq) Total Ionic Equation. In summary, covalent compounds are softer, have lower boiling and melting points, are more flammable, are less soluble in water and do not conduct electricity compared to ionic compounds. Since there are no spectator ions, the total ionic and the net ionic are exactly the same. Based on their positions, predict whether each solid is ionic, molecular, covalent, or metallic. Chemical Reaction can be expressed by: Molecular Equation (balanced chemical equation) Complete Ionic Equation (showing all ions in reaction) Net Ionic Equation (showing only those ions directly involved in reaction) Consider Slideshow. In the reaction of a weak acid and a weak base there is no spectator ion. A shared pair of e- is and anions (atoms that have gained e-).  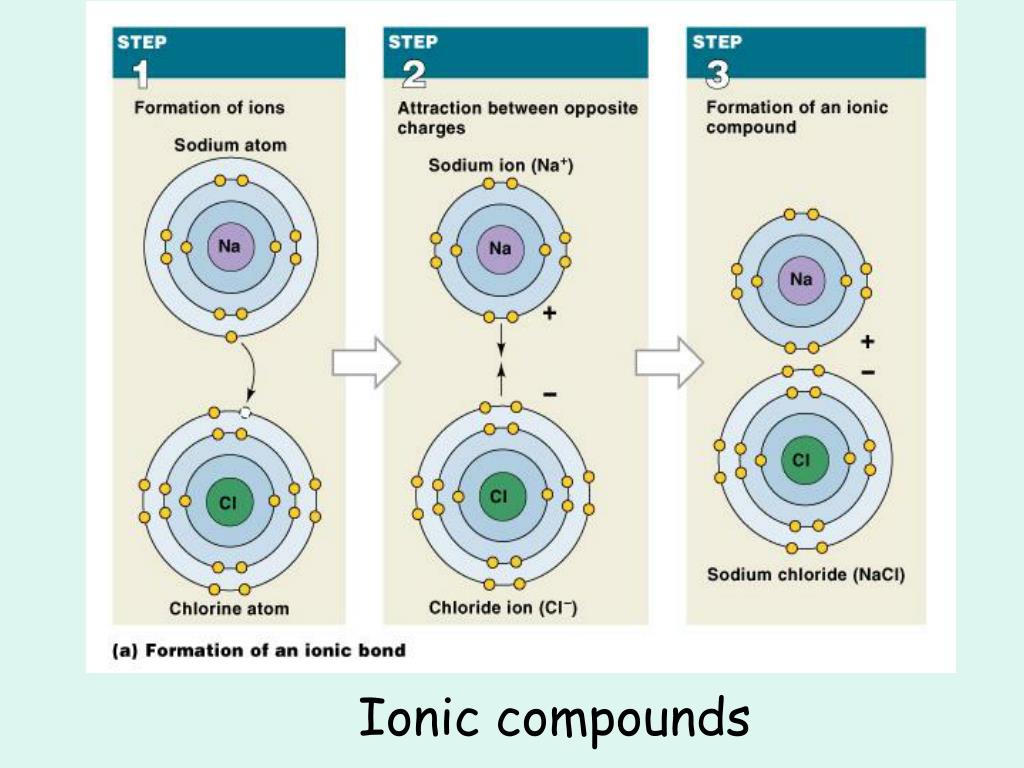 
Locate the component element(s) in the periodic table. Differences between Ionic and Molecular compounds Ionic compounds: Covalent or Molecular compounds: Are formed by electrostatic attraction Are formed by sharing of electron pairs in between cations (atoms that have lost e-) between atoms. Molecular Compounds GetChemistryHelp 20K subscribers Subscribe 1.6K Share Save 128K views 10 years ago Chemical Nomenclature.\) as ionic, molecular, covalent, or metallic solids and arrange them in order of increasing melting points.Īsked for: classification and order of melting points 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
